 source: Market department
source: Market department  date:2022-04-20
date:2022-04-20Gather your strength and create a new chapter in the industry. On April 13th, the online forum of "Big Charity Cloud Forum- -WHO Vaccine Pre-Certification (PQ)" jointly hosted by TIANS Group and Yunzhi Pharmaceutical Technology was held as scheduled. Mr.Christian Bachofen, an internationally renowned biomedical design and planning expert, director of The Group and chief technology officer of biomedicine, shared the special topic of "WHO Pre-certification Product Review Document Architecture and Good Document Specification". Nearly 3,000 professional guests of r & D, verification, quality management from biomedical enterprises across the country watched it online.
Mr.Christian Cloud Forum live course live
At present, a total of 7 Chinese enterprises have passed the WHO vaccine pre-certification, 5 of which are professional services provided by Mr.Christian, and they have rich practical experience in Chinese localization in the WHO-PQ field. At the same time, Mr.Christian explained the system document system of WHO pre-certification products in combination with his concept of serving international and Chinese pharmaceutical enterprises in nearly 20 years. He pointed out that WHO drug pre-certification is a service provided by WHO to evaluate the quality, safety and effectiveness of drugs, and explained the operation process mode of WHO pre-certification from seven steps, including the pre-submission meeting, application letter and pre-certification documents.

Mr.Christian shared the special topic of "WHO Pre-certification Product Review Document Architecture and Good Document Specification" in the cloud forum
First of all, we introduced the basic situation of WHO-PQ, he pointed out that the WHO drug pre-certification is a service provided by the WHO, used to evaluate the quality, safety and effectiveness of drugs, after he from the pre-submission meeting, application letter, pre-certification documents and other seven steps to explain the operation process mode of WHO pre-certification.
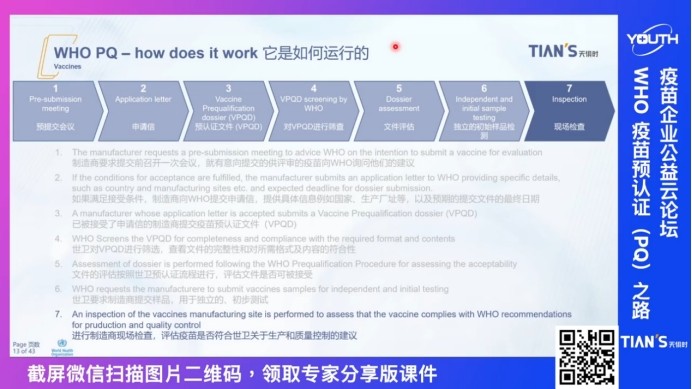
WHO pre-certification process mode explanation
Electronic general technical document (eCTD), as the current internationally recognized drug declaration data preparation format, has replaced the previous product master document (PSF). Mr Christian detailed the general technical documents (eCTD) in the specific content of the five modules, for the more important quality, the clinical trial report and clinical trial report the three plates, Mr Christian listed each plate need to write content and need to pay attention to the technical difficulties, for you to effectively solve the file writing questions and confusion.
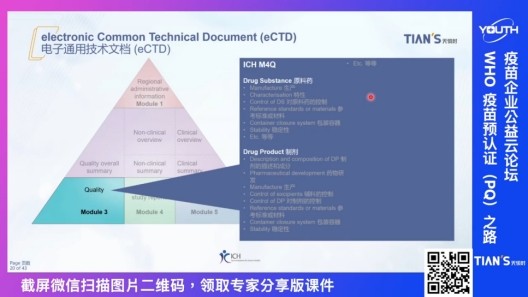
Explanation of eCTD Electronic General Technical Document course
Good document processing specification (GdP) is an indispensable part of the WHO pre-certification process. Mr.Christian started with an overall overview of how to record, data entry and recording notes, and the integrity and reliability of the data, and stressed that the more important aspect now is to ensure the source of information while ensuring the traceability and consistency of the document.
First appearance, connect to the world
In this online forum, we also released the "Pharmaceutical Professional English Vocabulary Manual" jointly produced by TIANS Group and Yunzhi Pharmaceutical Technology. The pocket book is classified by five parts: quality management (Quality Assurance), monitoring and analysis (Monitoring & Analysis), facilities (Facility & Equipment), production process (Production & Process), and registration and research and development (Registration & R & D), with a total of 2,000 + professional words. Each vocabulary includes Chinese, English, English phonetic symbols and vocabulary application examples. Under the wave of globalization of Chinese pharmaceutical enterprises, the Pharmaceutical Professional English Vocabulary Manual will also become a necessary professional "English dictionary" for pharmaceutical professionals.
Connect with the world and build the future of the industry. As the leader of pharmaceutical engineering EPC, The Group always takes science and technology as the source power of development, shares cutting-edge information in the pharmaceutical and environmental protection industries, spreads international cutting-edge ideas, and promotes the rapid development of the pharmaceutical industry.
Character link:ChristianBachofen

Mr.Christian Bachofen is an internationally renowned biomedical design and planning expert, with a bachelor in bioengineering at Zurich, and a founder and chief designer of CB Consultancy AG. He has served as a visiting professor at Basel and Jilin University in Switzerland. Mr.Christian has rich experience in the planning and design of pharmaceutical factory facilities, GMP clean room and HVAC system design, pharmaceutical water system design, quality management system, etc. As a project leader participated in several international famous biomedical design projects, including Johnson & Johnson pharmaceutical freeze-dried antibody production plant and mab-Xilin bottle packaging line design project, novartis pharmaceutical research and development park office building and for laboratory building design project, baekje shenzhou Suzhou pilot production workshop and Guangzhou commercial production workshop design project and sheng group new crown vaccine production workshop, and many other projects. The F2P2-Centocor's New R & D Fill Finish Pilot Plant project won the best project of the International Association of Pharmaceutical Engineering (ISPE). Christian In the design and implementation of the project, it proposed and realized a highly integrated unique design concept, cleverly balanced the requirements of flexibility, efficiency and compliance, and ISPE awarded it the best engineer.


 BOOK
BOOK 中文
中文
 Tender Hall
Tender Hall  Resource Center
Resource Center

 RETURN
RETURN